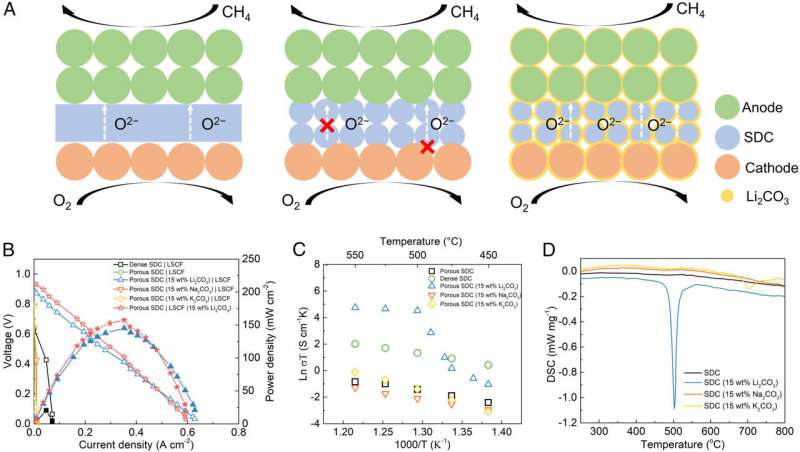
(A) Schematic of the conventional SOFC, porous SOFC, and the CSSFC. (B) The I-V-P performance of different fuel cell configurations with Ni-BZCYYb as anodes operated on CH4 at 550 °C. (C) The temperature-dependent Arrhenius plot of oxygen ionic conductivities of different electrolytes with or without carbonate modification. (D) DSC plots of different electrolytes in Ar atmosphere. Credit: Proceedings of the National Academy of Sciences (2022). DOI: 10.1073/pnas.2208750119
Like batteries, fuel cells produce energy through an electrochemical process. Unlike batteries, they don't run down or require recharging. However, the potential advantages of fuel cells are offset by challenges that include cost, performance and durability.
Michigan Technological University researcher Yun Hang Hu and two graduate students, Hanrui Su and Wei Zhang, took on those challenges, changing the conventional path of a fuel cell by creating an interface between the electrolyte and melted carbonate as an ultrafast channel for oxygen ion transfer.
"This allowed us to invent an entirely new type of fuel cell, a carbonate-superstructured solid fuel cell (CSSFC)," said Hu, who holds the Charles and Carroll McArthur Endowed Chair Professorship of Materials Science and Engineering in the Department of Materials Science and Engineering at Michigan Tech.
Like other fuel cells, CSSFCs have a wide array of potential uses, from providing energy to operate fuel cell vehicles and home power generation to entire power stations. Because CSSFCs are fuel flexible, they offer higher durability and energy conversion efficiency at lower operating temperatures than other types of fuel cells.
Most fuel cells are powered by hydrogen—typically produced from hydrogen-containing compounds, most often methane—via an expensive process called reforming. But the CSSFC developed in Hu's lab can directly use methane or other hydrocarbon fuels.
Hu said fuel flexibility is of particular interest for commercial applications. And, the new fuel cell's electrochemical performance at lower operating temperatures offers several other advantages. "The operating temperature of a conventional solid oxide fuel cell is usually 800 degrees Celsius or higher, because ion transfer in a solid electrolyte is very slow at a lower temperature," Hu said. "In contrast, the CSSFC's superstructured electrolyte can provide a fast ion transfer at 550 degrees Celsius or lower—even as low as 470 degrees Celsius."
The relatively low operating temperature offers high theoretical efficiency and lower cell fabrication costs. Hu said it is also potentially safer to operate than other solid fuel cells.
Tests on the CSSFC also showed an unprecedentedly high open circuit voltage (OCV), which indicates no current leakage loss and high energy conversion efficiency.
Hu estimates that CSSFC fuel efficiency could reach 60%. By comparison, the average fuel efficiency of a combustion engine ranges between 35% and 30%. The CSSFC's higher fuel efficiency could lead to lower carbon dioxide emissions in vehicles.
The research is published in the Proceedings of the National Academy of Sciences.
More information: Hanrui Su et al, Carbonate-superstructured solid fuel cells with hydrocarbon fuels, Proceedings of the National Academy of Sciences (2022). DOI: 10.1073/pnas.2208750119
Journal information: Proceedings of the National Academy of Sciences
Provided by Michigan Technological University